Rituximab in Multiple Sclerosis: B-lymphocyte population study and its relationship with dosing regimen
Abstract
Introduction:
Rituximab, a monoclonal antibody directed at CD20 positive B-lymphocytes, is used as an off-label treatment in MS, with good efficacy and safety profile. CD20-depleting strategies have a variable effect in the different B-cell subpopulations and the recovery after depletion determines therapeutic and safety profiles.
Our aim was to study the effectiveness and safety of rituximab in MS patients and their relationship with treatment regimen and B-lymphocyte population study.
Methods:
Retrospective observational single-center study of MS patients treated with rituximab for ≥6 months. Demographics, dosing regimen, B lymphocyte population study (6 and 12 months after treatment start) and adverse events were analysed.
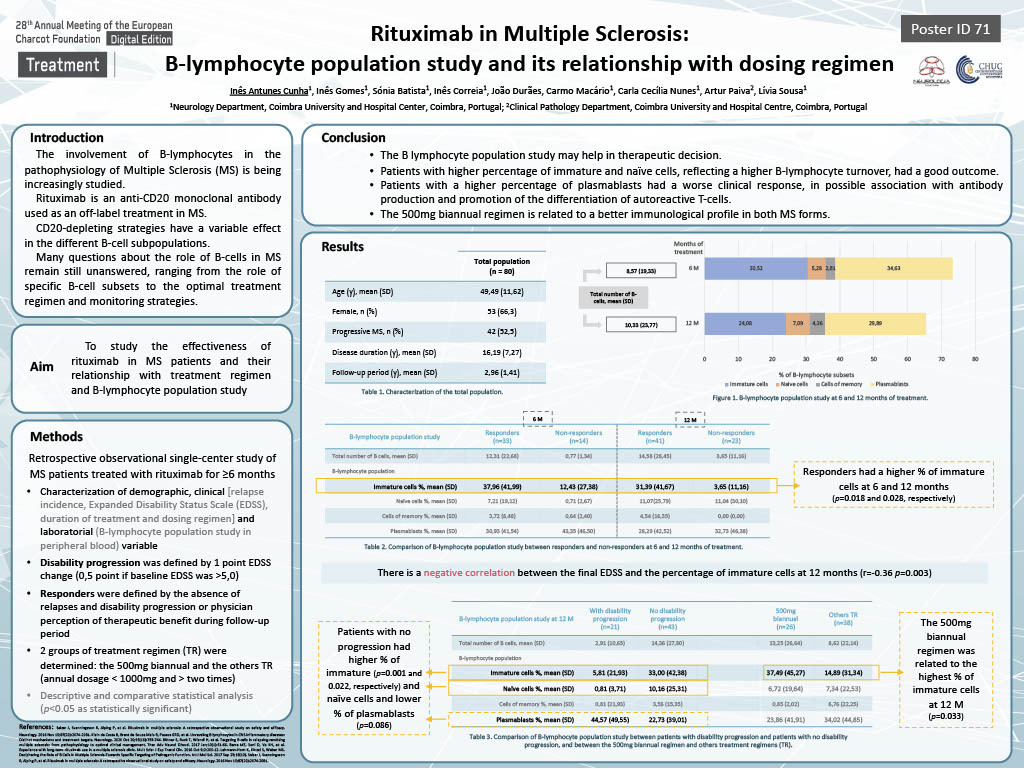
Results:
We included 80 patients, 53 (66.3%) women, with a mean age of 49,49 years (SD 11.62), 38 with relapsing-remitting MS (47.5%) and 42 with progressive MS (52.5%). At 6 months of treatment, the mean of total number of B cells was 8.57 (SD 19.33), corresponding to 30.52% immature B cells, 5.28% naive, 2.81% cells of memory and 34.63% plasmoblasts. At 12 months, there was a mean of 10.33 B cells (SD 23.77), with 24.08% immature cells, 7.09% naive, 4.36% memory cells and 29.89% plasmoblasts. Patients who responded to treatment had a higher percentage of immature cells at 6 and 12 months (p=0.018 and 0.028, respectively). The 500mg biannual regimen was related to the highest percentage of this lymphocyte subpopulation at 12 months (p=0.033). There is a negative correlation between the final EDSS and the percentage of immature cells at 12 months (r= -0.36, p=0.003).
Conclusion:
The B lymphocyte population study may help in therapeutic decision. Patients with lower percentage of immature cells, reflecting a lower B-lymphocyte turnover, had a worse clinical response. The 500mg biannual regimen is related to a better immunological profile in both MS forms.