Laboratory detection of antibody in IIDDs
Abstract
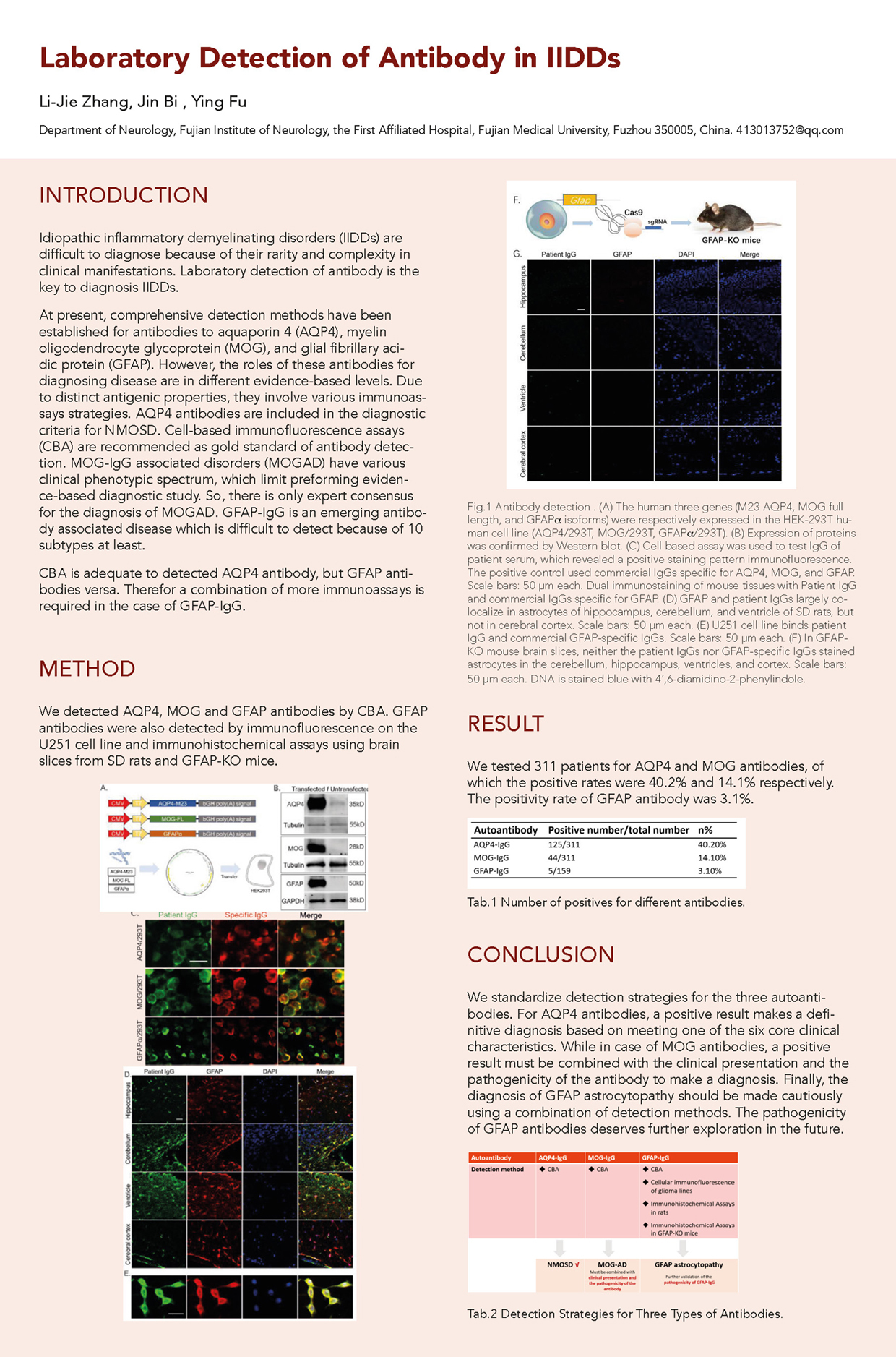
Idiopathic inflammatory demyelinating disorders (IIDDs) are rare and difficult to diagnose because of their variety and complexity of clinical manifestations. Laboratory detection of antibody is the key to the diagnosis of IIDDs. At present, we have established comprehensive tests for antibodies to aquaporin 4 (AQP4) , full length of myelin oligodendrocyte glycoprotein (MOG), and glial fibrillary acidic protein (GFAP) . The significance of these antibodies for the diagnosis of disease is inconsistent. They also involve a variety of immunoassay methods due to their different antigenic properties. AQP4 antibodies are included in the diagnostic criteria for NMOSD. The cell-based immunofluorescence assays (CBA) is recommended as the gold standard for antibody detection. MOG-IgG associated disorders (MOGAD) has clinical heterogeneity and is currently only an international consensus on the diagnosis and antibody testing, with no evidence-based medical evidence and no diagnostic criteria. GFAP antibody is gaining recognition as an emerging antibody. It is difficult to detect because there are at least 10 subtypes of GFAP. Therefore, we have detected AQP4 and MOG antibodies by CBA. We were able to cover almost all GFAP subtypes of antibodies by designing and constructing a variety of plasmids such as GFAPα, δ, κ, and +1 and then detecting them by CBA. The GFAP antibodies have been further verified by the rat brain slice immunohistochemical assays and U251 lineage cell immunofluorescence assays. Meanwhile, we have successfully constructed GFAP-KO mice using CRISPR/Cas9 technique and determined the specificity of GFAP antibodies by immunofluorescence of brain slices from GFAP-KO mice.
KEY WORDS: Idiopathic inflammatory demyelinating disorders (IIDDs), detection of antibody, GFAP-KO mice